Therapeutics
Aegros' Hyperimmune against Covid-19
Aegros is developing Covimmune™, a hyperimmune against Covid-19, as our first product.
Extensive research and clinical trials are being undertaken because it is vital that this new treatment option against Covid-19 takes its place amongst the medicines that have already been developed. This is because existing treatments have tended to wane in effectiveness over time. Further, there are some patients, particularly those who are immuno-compromised, who cannot benefit from existing therapies.
The video above demonstrates how Aegros' Covimmune™ can keep pace with an elusive virus, maintaining its effectiveness even as the virus changes.
Traditional Plasma Derived Medicinal Products
Aegros' Hyperimmune against Covid-19
Aegros is developing Covimmune™, a hyperimmune against Covid-19, as our first product.
Extensive research and clinical trials are being undertaken because it is vital that this new treatment option against Covid-19 takes its place amongst the medicines that have already been developed. This is because existing treatments have tended to wane in effectiveness over time. Further, there are some patients, particularly those who are immuno-compromised, who cannot benefit from existing therapies.
The video above demonstrates how Aegros' Covimmune™ can keep pace with an elusive virus, maintaining its effectiveness even as the virus changes.
Traditional Plasma Derived Medicinal Products
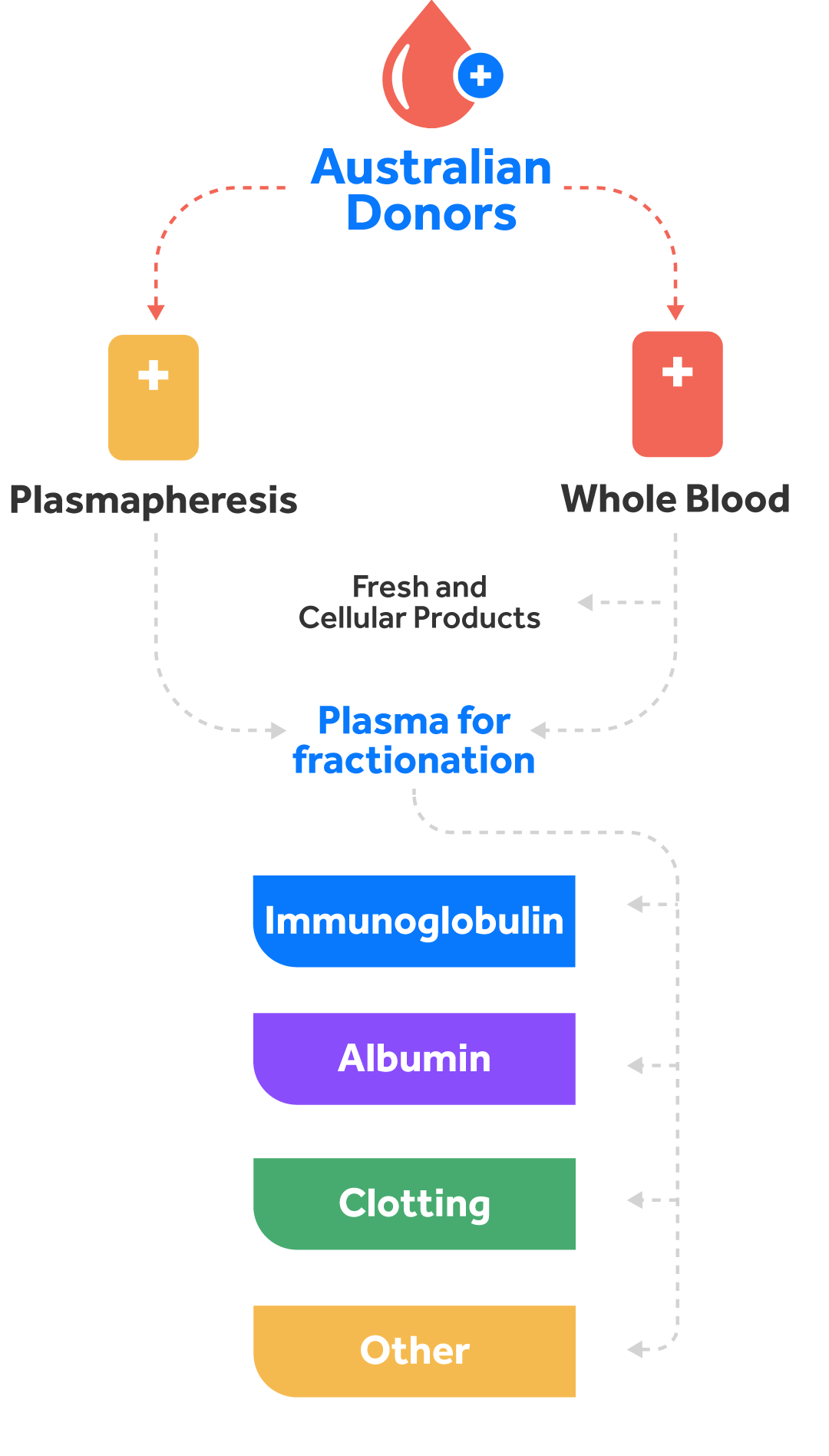
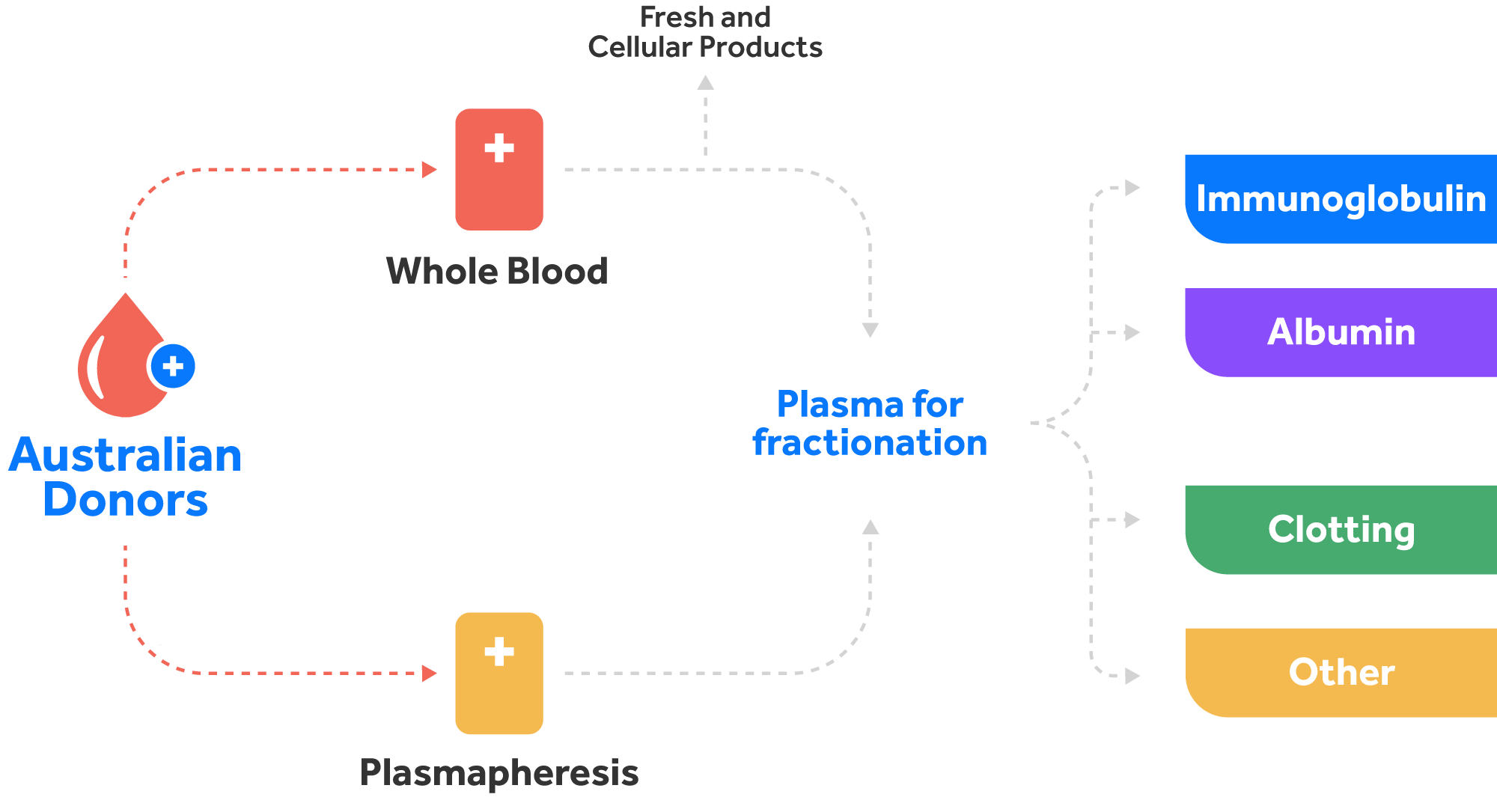
Fractionation of human plasma remains the only approach to make several life-saving therapies required to treat a number of human diseases.
In Australia, we rely on voluntary, non-remunerated donations for blood and blood products, and only import blood products where needed. Each year ~3% of Australians donates blood (whole blood) and plasma (via plasmapheresis). According to Australian Red Cross Lifeblood, about 1 in 3 of us will need blood or blood products at some stage in our lives to treat existing conditions or following an emergency.
Blood contains approximately 55% plasma. Plasma is the clear to straw coloured liquid portion of blood that remains after red cells, white cells and platelets are removed. Plasma has multiple components with different clinical uses, and it cannot be manufactured in a laboratory.
A range of valuable health products such as immunoglobulins (Ig), clotting factors, albumin and others are manufactured from blood plasma through a process called fractionation, in which different types of proteins found in blood plasma are separated, purified, and concentrated into therapeutic doses.
The COVID-19 pandemic has demonstrated the fragility of our healthcare systems and the need for rapid development of effective novel therapies. The risk of COVID-19 variants and new epidemics must be addressed.

Aegros has commenced operations with production of a hyperimmune intravenous antibody targeted against SARS-CoV-2. This product may be of use to people in whom vaccines are not entirely effective e.g. those living with severe immunocompromise.
As Australia’s newest fractionator, Aegros intends to manufacture a full suite of plasma therapeutics. With its significant R&D capability and unique fractionation process HaemaFrac®, Aegros is looking to develop, in conjunction with Australia’s medical community, novel plasma proteins to address diseases with unmet therapeutic needs.