

Aegros was proud to attend and sponsor the 32nd Annual Conference of the Australasian Society of Clinical Immunology and Allergy, held Aug 30 – 2 September in Melbourne. An Aegros and Kirby Institute research paper was featured at the conference, and the poster and abstract for this paper are available below.
ABSTACT:
ANTI-SARS-COV-2 HYPERIMMUNE GLOBULIN FROM CONVALESCENT PLASMA: ACTIVITY & POTENTIAL APPLICATIONS
Authors:
Guy Gavagna1, Stephen Mahler1,2, Stuart Turville3, Anthony Kelleher3, Hari Nair1,2
1 Aegros Ltd, Macquarie Park, Australia
2 University of Queensland, St Lucia, Australia
3 Kirby Institute, University of New South Wales, Kensington, Australia
INTRODUCTION:
COVID-19 has caused significant morbidity and mortality, especially for those with immunocompromise. Unfortunately, increased immune evasion from SARS-CoV-2 variants of concern (VOC) has diminished the effectiveness of vaccines, monoclonal antibodies and antivirals. Further, many with immunocompromise remain serosilent despite repeated vaccination against SARS-CoV-2[1]. The search continues for therapies with higher barriers to viral resistance. Hyperimmune immunoglobulin (HIG) has a long history of use as passive immunity against viruses[2]. In COVID-19, HIG blocks SARS-CoV-2 binding to ACE2 and inactivates SARS-CoV-2[3]. Early administration of high titre COVID-19 convalescent plasma (CCP) reduces risk of disease progression in patients with COVID-19[4]. Broadly specific anti-SARS-CoV-2 antibodies in CCP, present at greater concentrations in HIG, have demonstrated neutralising activity against a range of VOC[5].
METHOD:
Pooled CCP collected in January 2022 was fractionated (Aegros Ltd, Sydney) using the novel HaemaFrac® process to produce HIG complying with European Pharmacopoeia (EP) for human normal Ig for intravenous administration. Independent analyses of pooled plasma for manufacture and final HIG product are reported here.
RESULTS:
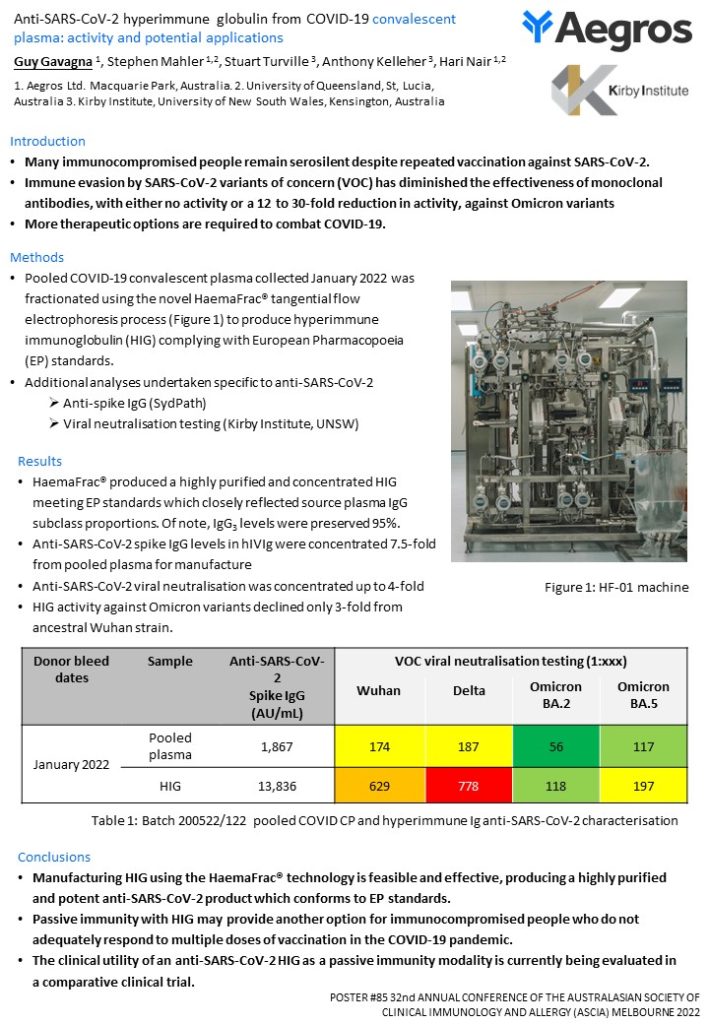
HaemaFrac® produced a highly purified and concentrated anti-SARS-CoV-2 HIG meeting EP standards. IgG subclass proportions closely reflected source CCP: IgG1: 65.5% (57.3%), IgG2: 26.1% (30.8%), IgG3: 6.9% (7.3%) and IgG4: 1.5% (4.6%). Anti-SARS-CoV-2 IgG levels were concentrated 7.5-fold from CCP. Compared with starting pooled CCP, HIG viral neutralisation titres against Wuhan and VOCs Beta, Delta and Omicron-BA.5 were increased up to 4-fold.
CONCLUSION:
The HaemaFrac® technology produces HIG with highly conserved levels of IgG3, concentrated levels of anti-SARS-CoV-2 IgG, and up to 4-fold increase in neutralisation capacity against ancestral and subsequent VOC. Neutralising antibodies are an important correlate of protection against SARS-CoV-2[6], and IgG3 possesses anti-viral effector functions,[7],[8]. Therefore an anti-SARS-CoV-2 HIG may provide therapeutic advantages for immunocompromised people with inadequate endogenous antibodies levels in response to vaccination.
POSTER:
REFERENCES:
[1] International Society of Blood Transfusion (ISBT) 2022- What’s new in COVID. Accessed June 10, 2022
[2] Richard L. Wasserman, Chapter 55 – Immunoglobulin replacement, Editor(s): Kathleen E. Sullivan, E. Richard Stiehm, Stiehm’s Immune Deficiencies (Second Edition), Academic Press, 2020, Pages 1143-1156
[3] Focosi D, Franchini M, Tuccori M, Cruciani M. Efficacy of high dose polyclonal intravenous immunoglobulin in COVID-19: A systematic review. Vaccines (Basel). 2022;10(1):94.
[4]Sullivan DJ, Gebo KA, Shoham S, et al. Early outpatient treatment for Covid-19 with convalescent plasma. N Engl J Med 2022;386:1700-11.
[5] Li M, Beck EJ, Laeyendecker O, et al. High Viral Specific Antibody Convalescent Plasma Effectively Neutralizes SARS-CoV-2 Variants of Concern. Preprint. medRxiv. 2022;2022.03.01.22271662. Published 2022 Mar 2. doi:10.1101/2022.03.01.22271662
[6] Stadler E, Li Chai K, Schlub T E, Cromer D et al. Determinants of passive antibody effectiveness in SARS-CoV-2 infection. medRxiv March 2022 preprint
[7] Vidarsson G, Dekkers G and Rispens T (2014) IgG subclasses and allotypes: from structure to effector functions. Front. Immunol.5:520. doi:10.3389/fimmu.2014.00520
[8] Kober C, Manni S, Wolff S, Barnes T, Mukherjee S, Vogel T, et al. (2022) IgG3 and IgM Identified as Key to SARS-CoV-2 Neutralization in Convalescent Plasma Pools. PLoS ONE 17(1): e0262162.